Strep A Rapid Test Device Cassettes with Controls Box/20 Tests
The Rapid Response Strep A Rapid Test Device is a rapid test for the presumptive detection of Group A Streptococcus antigens in human throat swab specimens. This kit is intended for use as an aid in the diagnosis of Strep A infection directly from throat swabs to help diagnose and administer therapy immediately.
- Rapid test for detecting Strep A infection.
- Controls included in kit.
- Includes all materials needed to perform the test.
- Comes with a sturdy, plastic tube stand for mess-free workspace.
- Read results in 5 minutes.
- No specialized training or equipment required.
- Uses easy-to-collect throat swab specimens.
- No cross-reactivity with 53 common respiratory tract organisms.
- For point-of-care in vitro diagnostic use only.
Features
- Read results in 5 minutes
- Detects Strep A infection
- Throat swab sample collection
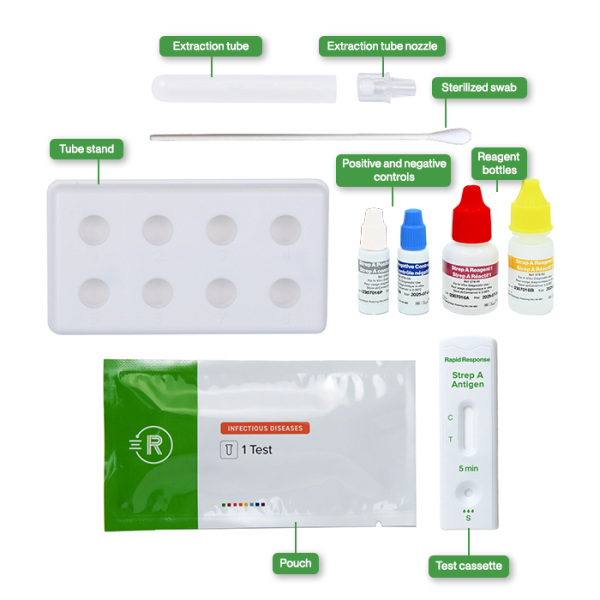
- 20 Individually packed test devices
- 1 Bottle of Reagent 1
- 1 Bottle of Reagent 2
- 20 Sterilized swabs
- 20 Nozzles with filters
- 20 Extraction tubes
- 1 Tube stand
- 1 Positive control
- 1 Negative control
- Package insert
What is Strep A
Beta-hemolytic Group A Streptococcus is a major cause of upper respiratory infections such as tonsillitis, pharyngitis, and scarlet fever. Early diagnosis and treatment of Group A Streptococcal pharyngitis has been shown to reduce the severity of symptoms and further complications, such as rheumatic fever and glomerulonephritis.
Conventional methods for detecting Strep A infection are dependent on isolation and subsequent identification of the organism, and often require 24-48 hours. Recent development of immunological techniques to detect Group A Streptococcal antigen directly from throat swabs allow physicians to diagnose and administer therapy immediately.
How accurate is it?
Diagnostic Sensitivity: 97.4% (93.6% ~ 99.0%)*
Diagnostic Specificity: 99.4% (96.8% ~ 99.9%)*
Overall Agreement: 98.5% (96.5% ~ 99.3%)*
*Clinical study from product insert.
Result interpretation
See picture
Please refer to the product insert for more details.
Proficiency testing from AAB and CAP
The BTNX Rapid Response Tests undergo regular evaluation using proficiency samples from AAB (American Association of Bioanalysts) and CAP (College of American Pathologists).